|
The ChemSpider ID of Chlorine can be noticed as 22933. CAS refers to Chemical Abstracts Service, which provides chemical information about chemical elements with unique identification. The CAS Identification number of Chlorine is. This is represented by following Aufbau principle by mentioning the stable configuration of Chlorine’s respective Noble gas in the same period. Chlorine electron configurationsĮlectronic configuration of Chlorine atom is obtained as Is 2 2s 2 2p 6 3s² 3p⁵. It means the atom can gain one electron but leave its 7 valence electrons and show different oxidation states. Chlorine oxidation statesĬhlorine shows many oxidation states or numbers such as −1, +1, +2, +3, +4, +5, +6, and +7. It is highest within series of ionisation energy as eliminating third atoms from the valence shell is not likely within Cl 2+ ion with electron urge. Third ionisation energy level of Choline atoms has been identified as 3822 kJ/mol. After leaving one electron, the atom becomes Cl + that demands more energy to become Cl 2+. This is higher than first one because atoms loses tendency of leaving the second atom. The second ionisation energy level of Chlorine atom is 2298 kJ/mol. This refers to the amount of energy requires in one Cl atom for eliminating one electrons from last electron shell or valence shell. The energy of first ionisation required for Chlorine has been obtained as 1251.2 kJ/mol. In the first shell, it has 2 electrons, in the second it possesses 8 electrons which is referred as a filled shell and the last one has 7 electrons. There are three-electron shells inside a Chlorine atom. Let us shortly discuss about electronic shell of Chlorine below. Chlorine electronic shellĮlectronic shell the electron-holding spaces inside an atom. 36Cl is a stable radioactive isotope, which has half-life of almost 30,000 years. Three main stable isotopes of Chlorine are 35 Cl, 36Cl, and 37Cl. Let us list out the discovered isotopes of Chlorine below. Isotopes are the types of atom differ by the number of nucleons present in its nucleus. This radius is noticed to differ between 98 pm to 108 pm depending one the different circumstances. Chlorine ionic/covalent radiusĬhlorine’s covalent radius has been identified as approximately 102pm. This radius is denoted by r w and it refers to the half of the distance between two single atom of Chlorine is 175 pm within a balance electrostatic force. Chlorine Van der Waals radiusĬhlorine has 175 pm of van der Waals radius. This boiling point is also noticed to be lower than other elements but higher than Fluorine. Boiling point of Chlorine has been recognised as −34.04 ☌ (239.11 K, −29.27 ☏). It can be said that the meting point of Cl is very low but higher than the same of Fluorine. The melting point of Chlorine is noticed to be −101.5 ☌ (171.6 K, −150.7 ☏). At STP, standard amount of atomic density is 3.2 g/L. The atomic density of Chlorine has been obtained as 3.214g/cm3. In Pauling scale of electronegativity, the electronegativity of Chlorine has been measures by 3.16. 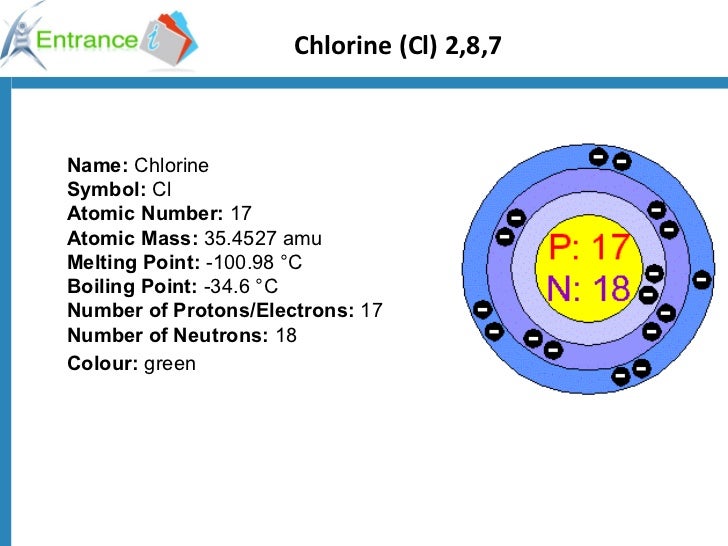
Chlorine Electronegativity according to Pauling Chlorine atomic number Chlorine atomic WeightĪtomic weight of Chlorine has been calculated as approximately 35.453 u. This means that Chlorine has 17 protons and 17 electrons in a single atom. Chlorine atomic numberĬhlorine is noticed to have atomic number of 17. Chlorine block in periodic tableĬhlorine has been placed at d-block by Mendeleev, who was the former of the modern periodic table. This period of selection of Chlorine has been done by the number of electronic shell present in its atom. Chlorine period in periodic tableĬhlorine can be identified in period 2 in the periodic table. Therefore, it can be said that groups are the columns in periodic table. Chlorine group in periodic tableĬhlorine is placed at group 17 where all the other Halogen elements are placed vertically. As ‘C’ and ‘l’ are emphasised in the word ‘Chlorine’, these two letters is used as its symbol. “Cl” is used as symbol of Chlorine in chemistry to represent it in chemical reactions. All the relevant facts such as meting point, boiling point, electronegativity, ionisation energies and others will be discussed throughout this article. The chemical facts of the element are reliable in keenly examining the internal facts and chemical behaviour of element in chemical reaction with others.Ĭhemical properties of Chlorine including its position in the periodic table would be described in this article. It is the second halogen element in the periodic table. 
Let us continue describing the chemical properties of Chlorine below.Ĭhlorine chemical properties at first include the behaviour of Chlorine elements as a halogen. Chlorine chemical properties cover some relevant chemical facts about the periodic element Chlorine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
